thiophene) into their corresponding carbaldehydes - in this reaction, the proton of DMF is less acidic than that of a thiophene or proton exchange would occur in place of nucleophilic attacks. Note that the sequential treatment with butyllithium and DMF is a method to convert electron-rich aromatic rings (e.g. A much more likely deprotonation would be that of a methyl group which would extend the π system. While it might lead to a carbene, the carbene is not exactly stable. That proton is not even mentioned on both the Evans table and the Bordwell data. Obviously, there is no $\mathrm$ bond in DMF has a very, very low tendency to deprotonate. But let me note a couple of things first: This is a common attempt to visualise what π systems actually mean for the overall Lewis structure. What gives?Īlso, as a side question - the reason why the solvent is polar aprotic, despite having the capacity to shed a hydrogen from the carbon to form an induction-stabilized carbanion, is because protic solvents have the labile hydrogen attached exclusively to an oxygen or a nitrogen, correct? It says to base the answer off of part d), but there are two filled orbitals! How do I know which pair of electrons occupies which orbital?ĭoes the sp3 lone pair of the nitrogen occupy the lowest occupied MO or the highest occupied MO? Because it appears from the correct answer that it occupies the higher one - but why is that? The other delocalized pair of electrons is the pi electrons in the C=O double bond, and I get that maybe they have different energies, but I can't think of any reason one pair should occupy one particular orbital over the other.įurthermore, according to the movement of electron pairs in the resonance structures of f) (which were all correct according to the answer key), the third structure suggests the lone N pair is shared between the carbon and the nitrogen, not the oxygen and the nitrogen. 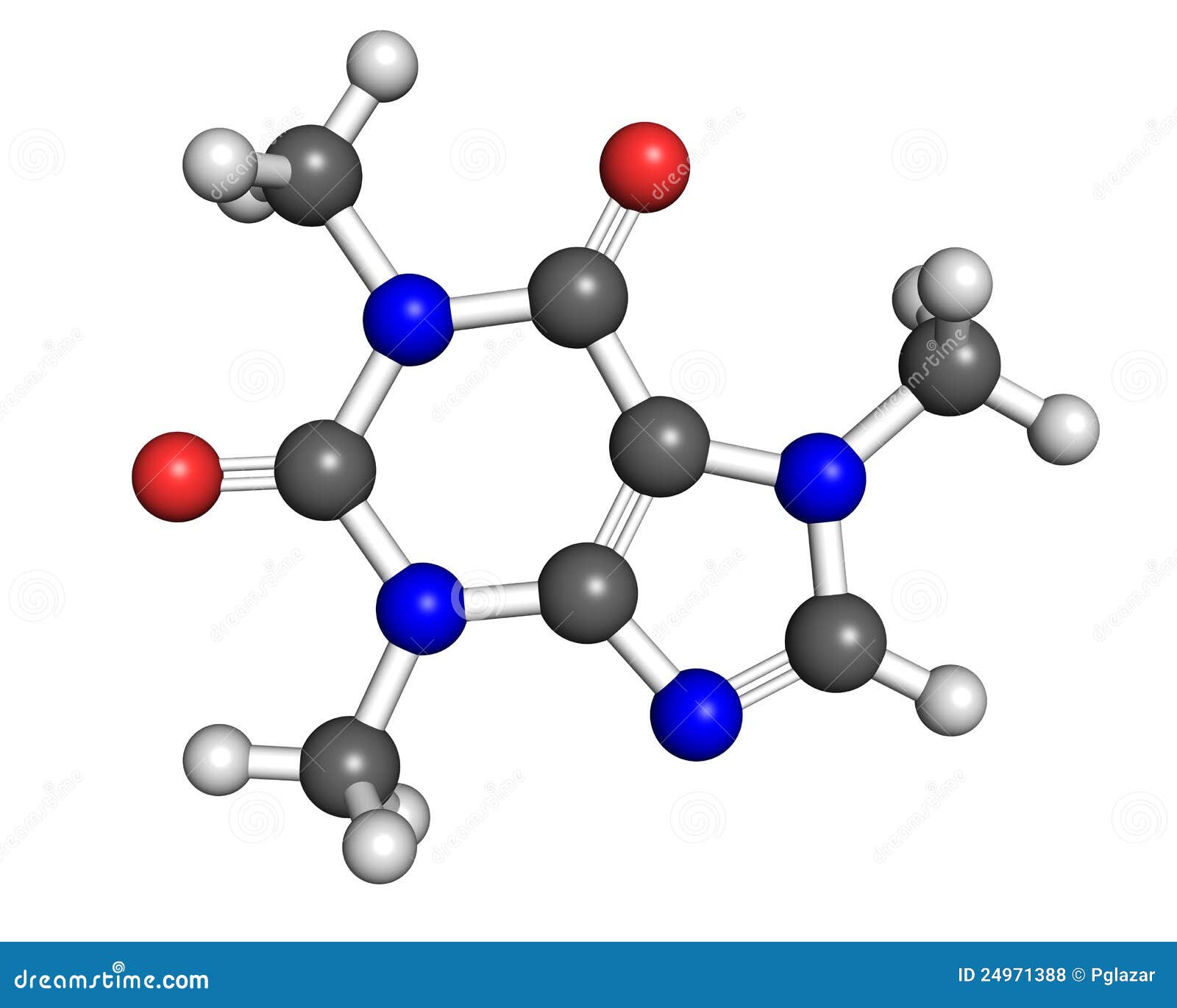
CO bond contains one sigma bond and one pi bond. 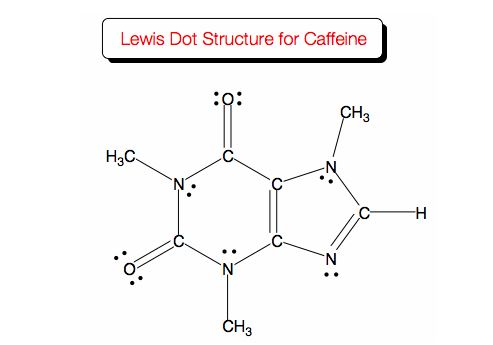
I was flitting back and forth between O and N, and O, N, and C, but in the end got it wrong. four pi bonds are present in caffeine and there are 24 sigma bonds present. I wasn't able to find anything about this online, either. The answers were posted, but there are no explanations for anything. The electrons of both lone pairs are not delocalized.I was just doing some practice midterms and I came across this question. The electrons of one lone pair are delocalized. The electrons of both lone pairs are delocalized. For each structure, answer the following questions: Are the lone-pair electrons delocalized? Is the structure aromatic? The lone pair is delocalized. The compound is aromatic not aromatic aromatic not aromatic aromatic not aromatic aromatic not aromatic Show transcribed image text Each cyclic structure shown below contains a heteroatom that has lone-pair electrons. 
The electrons of both lone pairs are not delocalized. Do not draw the wrong structure as it can cause errors in determining the location of lone pairs. The compound contains nitrogen, carbon, hydrogen and oxygen. For each structure, answer the following questions: Are the lone-pair electrons delocalized? Is the structure aromatic? The lone pair is delocalized. Step-by-step Step 1 of 3 Part a Structure of caffeine is given below: Caffeine is bicyclic in which two rings are fused, one ring is six-membered ring and another ring is five-membered ring. Each cyclic structure shown below contains a heteroatom that has lone-pair electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
